CaCO3: Definition, Preparation, Uses and Examples
By A Mystery Man Writer
Description
Calcium carbonate is one of the most known chemicals. Calcium carbonate is a non-toxic and odourless compound. It is found as a white mineral that occurs naturally in chalks, limestones and marbles. It is a common substance present in rocks as the mineral’s calcite and aragonite. CaCO3 is most notably limestone.

Preparation of High-Purity Calcium Carbonate by Mineral

Controlling the Vaterite CaCO3 Crystal Pores. Design of Tailor
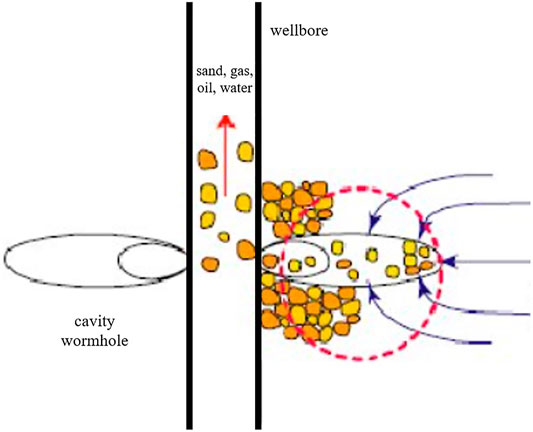
Frontiers A Review of Enzyme-Induced Calcium Carbonate

Monitoring of CaCO3 Nanoscale Structuration through Real-Time

CaCO3 crystals as versatile carriers for controlled delivery of
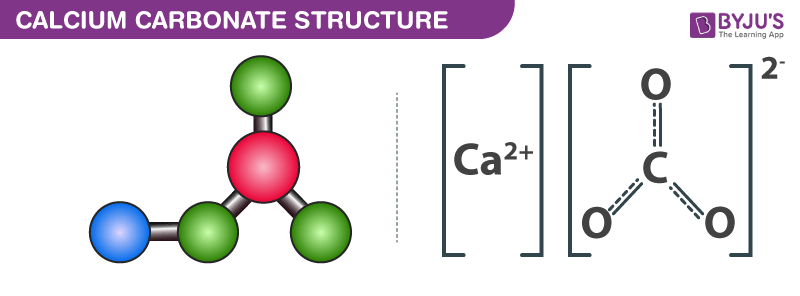
Limestone: Calcium Carbonate (CaCO3) - Uses, Preparation

Calcium carbonate - Wikipedia
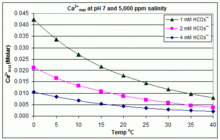
Calcium carbonate - Wikipedia

Daily use of Calcium Carbonate. The reaction between calcium

Calcium Carbonate(CaCO3) - Limestone Formula, Structure, Uses
.png)
What are Bases? - Definition, Examples, Types, Properties and Uses

Diagram of the CaCO 3 precipitation process by the liquid-liquid
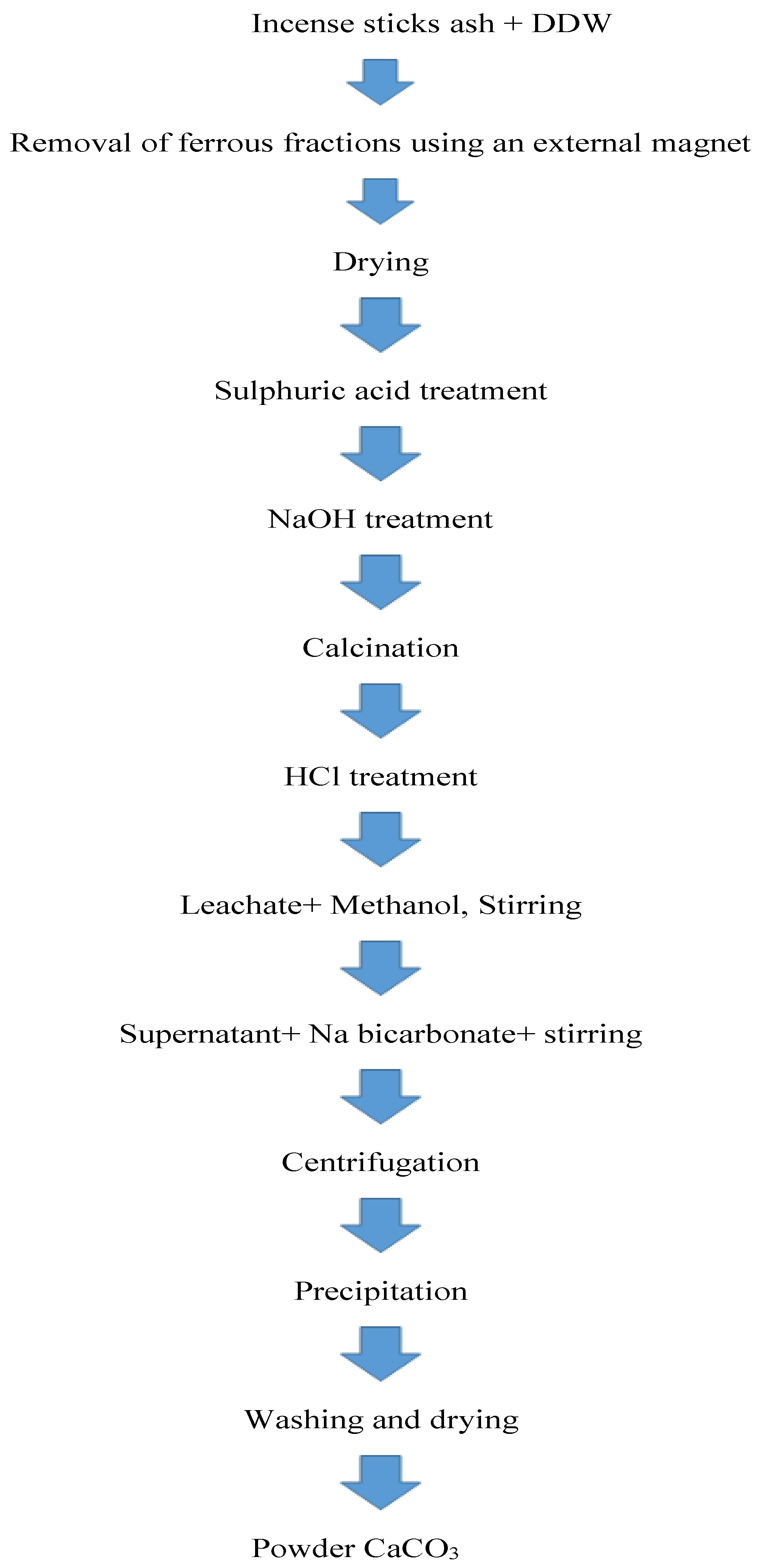
Applied Sciences, Free Full-Text
from
per adult (price varies by group size)






